📋 Product Description
FOR VETERINARY USE ONLY
COMPOSITION
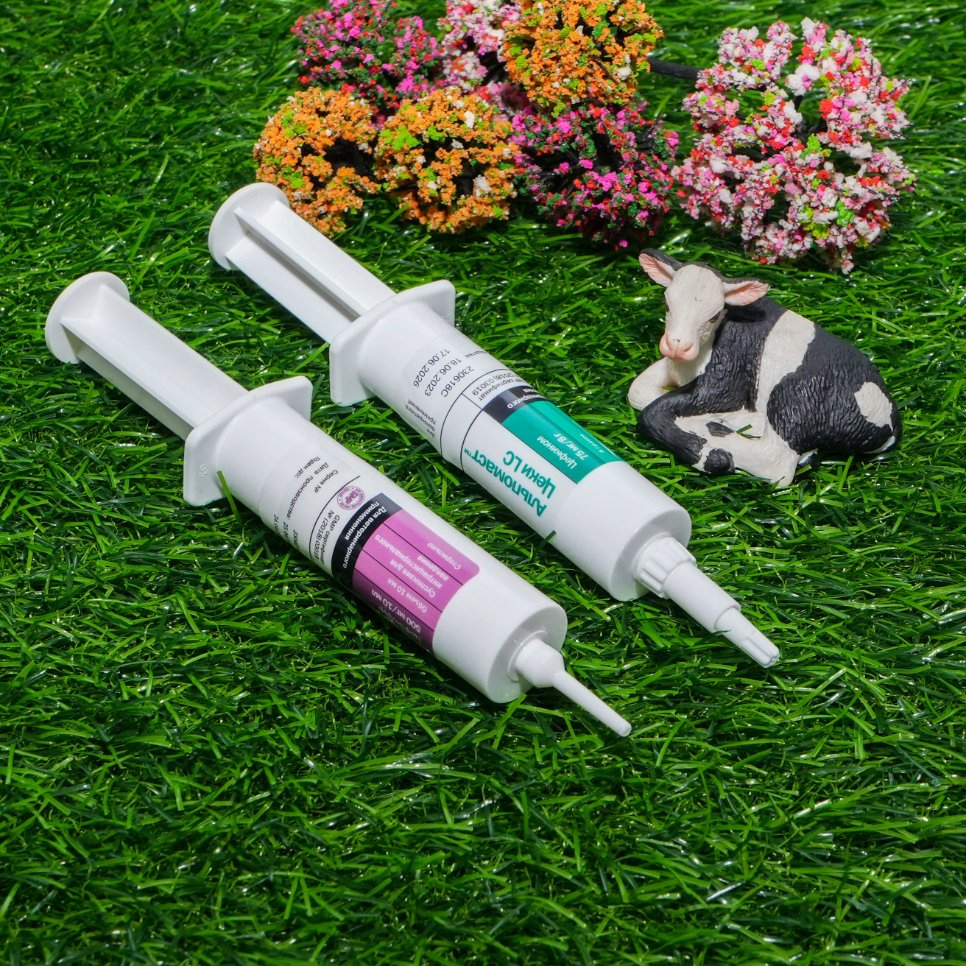
Each syringe contains cefquinome 900mg.
INDICATIONS
For the treatment of acute and chronic endometritis in dairy cows caused by sensitive bacteria.
DOSAGE AND ADMINISTRATION
Shake well before use. A rectal massage should be performed to remove the lochia before use, clean and disinfect the vaginal orifice and the perineum. Intrauterine perfusion, one syringe for a single dose. According to the condition, additional dose after 72 hours.
ADVERSE REACTION
No adverse reaction to follow the recommended dosage.
SPECIAL WARNINGS
Animals that are allergic to cephalosporins or other beta-lactam antibiotics are prohibited from using this product. Do not touch this product if you are allergic to penicillin and cephalosporin antibiotics. Shake well before use.
WITHDRAWAL PERIOD
Milk: 7 days.
STORAGE
Seal and store in a cool and dark place.
SHELF LIFE
3 years.
🏭 Professional Manufacturing
📜 Certifications
• Veterinary Medicine GMP Certificate & Production License
• ISO9001 Quality Management System
• ISO14001 Environmental Management System
• ISO45001 Health Management System
• Recognized Model Unit for International Cooperation
❓ Frequently Asked Questions
Q Are you a professional manufacturer?
Yes, we are a comprehensive high-tech enterprise specialized in R&D, production, and technical service for veterinary drugs and feed additives, established in 2001.
Q Do you hold GMP certification?
Yes, our facility successfully obtained its first GMP certification in 2005, with the latest update completed in 2022 to meet modern standards.
Q How do you ensure product quality?
Quality management is our focus. Our QC central laboratory is equipped with advanced inspection instruments. Professional inspectors with years of experience monitor every stage of production to ensure only qualified products reach our clients.
Q Which international standards do you comply with?
We hold a valid Manufacturing License and GMP certificate. Additionally, we are certified under ISO9001 (Quality), ISO14001 (Environment), and ISO45001 (Health & Safety).
Q Can you provide documents for product registration?
Yes, our professional registration team can provide necessary documentation including FSC, COPP, COA, Analysis Methods, Stability Reports, and Instructions.
Q Is it possible to visit your production site?
We welcome international customers to visit our factory. Many foreign clients visit us annually to inspect our veterinary medicine production lines.



 Vetsys Pharmaceutical
Vetsys Pharmaceutical