
1 / 5
 Vetsys Pharmaceutical
Vetsys Pharmaceutical










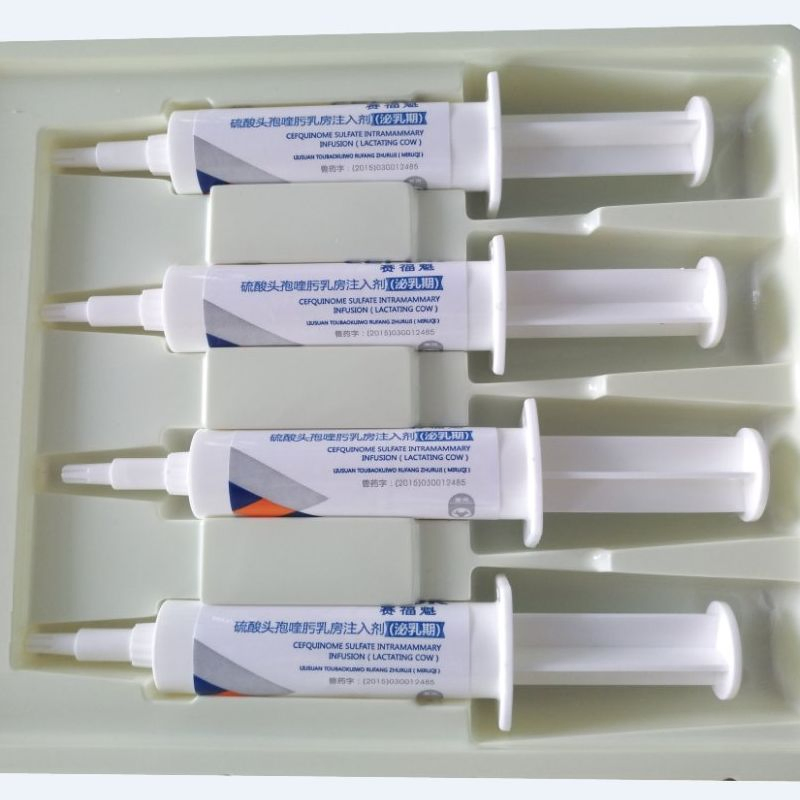
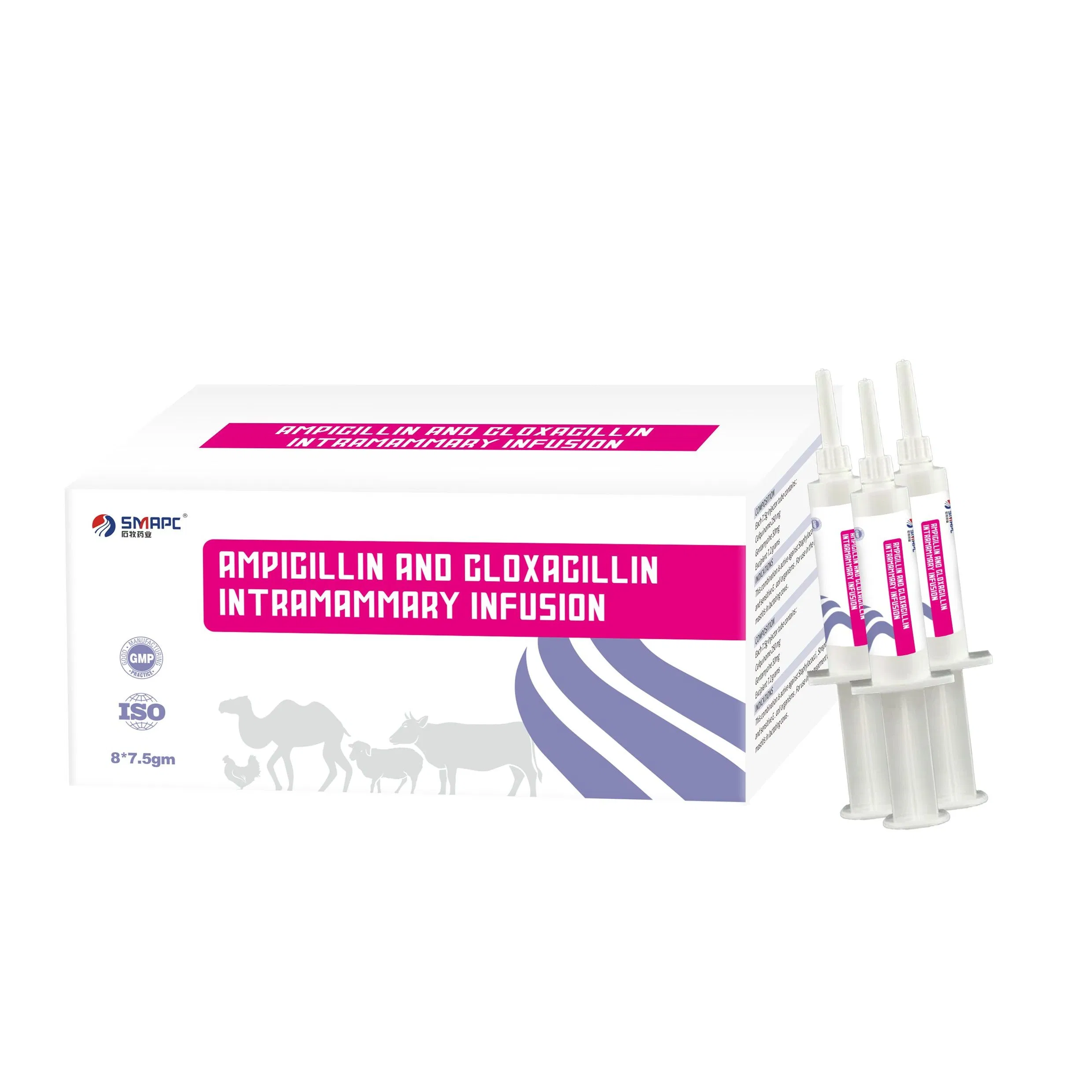
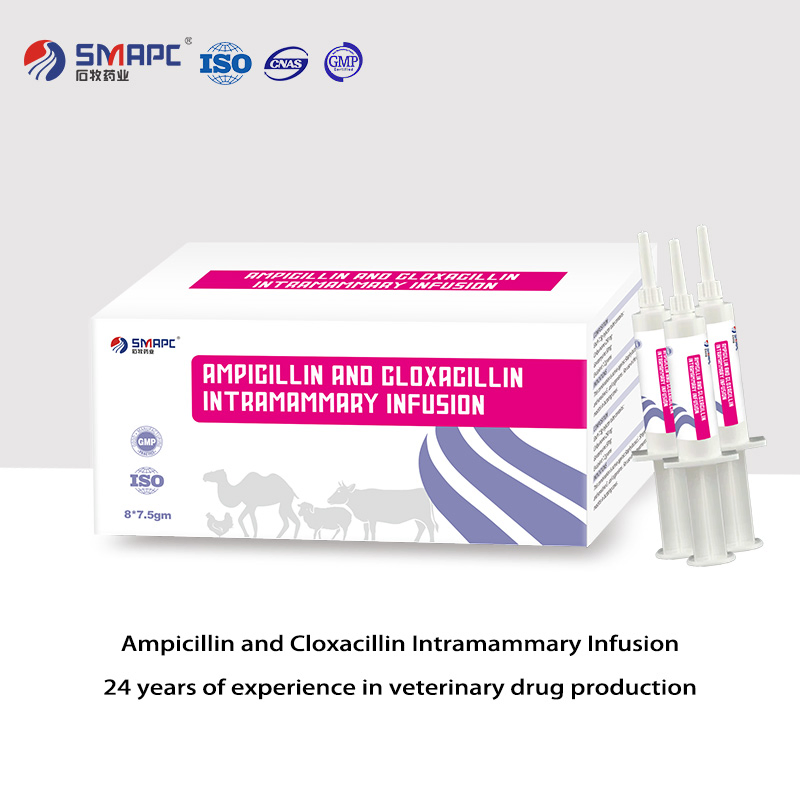
| Product Name | Ampicillin and Cloxacillin Intramammary Infusion |
| Model NO. | Ampicillin and Cloxacillin Intramammary Infusion |
| Pharmacodynamic Factors | Animal Species |
| Efficacy | Antibacterial |
| GMP / ISO | Yes / Certified |
| Specification | 100ml/bottle |
| Production Capacity | 50000 Pieces/Day |

| Withdrawal Period | Cow: 28 days; Milk: 60 hours (48 hours abandoned milk). |
| Storage | Seal and store in a cool, dark place (below 30ºC). |
| Shelf Life | 2 Years |



• Veterinary Medicine GMP Certificate & Production License
• ISO9001 Quality Management System
• ISO14001 Environmental Management System
• ISO45001 Health Management System









