Are you a manufacturer?
Yes, we are a professional high-tech enterprise established in 2001, specialized in R&D, production, marketing, and technical services for veterinary drugs and feed additives.
Do you have GMP certification?
Yes, we successfully obtained our first GMP certification in 2005, and our latest GMP standards were updated in 2022 to ensure the highest production quality.
How do you control product quality?
Quality is our focus. We maintain a Quality Assurance (QA) and Quality Control (QC) Central Laboratory equipped with advanced instruments. Our experienced inspectors monitor every stage to ensure no unqualified materials enter the process.
What management systems do you have in place?
Beyond GMP, we have passed certifications for ISO9001 (Quality), ISO14001 (Environmental), and ISO45001 (Health Management Systems).
Can you provide documents for registration?
Our professional registration team can provide necessary documentation including FSC, COPP, COA, Analysis Methods, SMF, Stability Reports, and Instructions.
Is factory visitation possible?
We welcome international customers to visit our pharmaceutical production facilities to witness our quality standards and production scale firsthand.
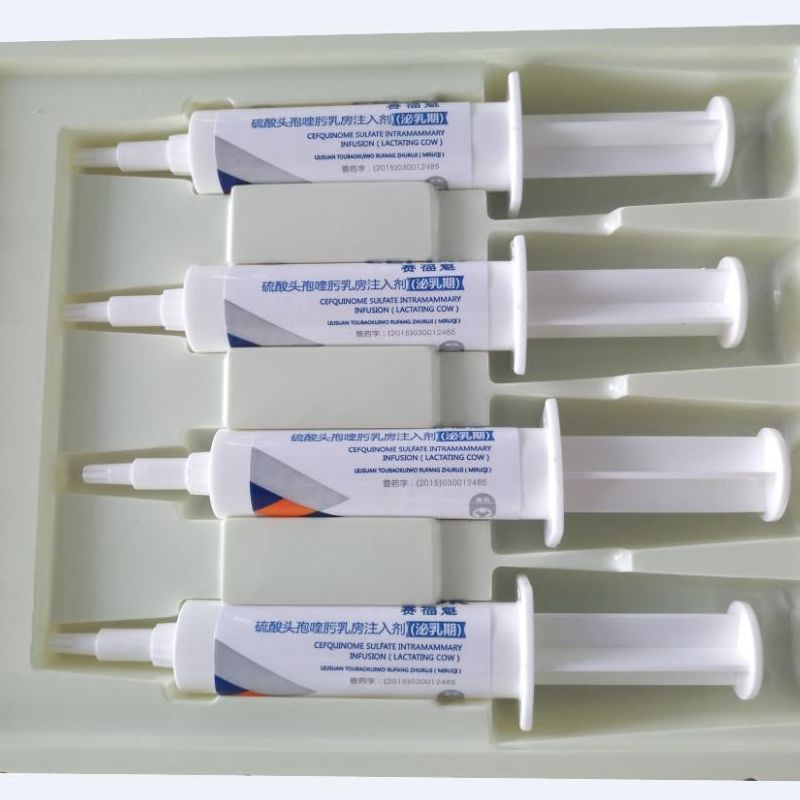


 Vetsys Pharmaceutical
Vetsys Pharmaceutical