


1 / 5
 Vetsys Pharmaceutical
Vetsys Pharmaceutical










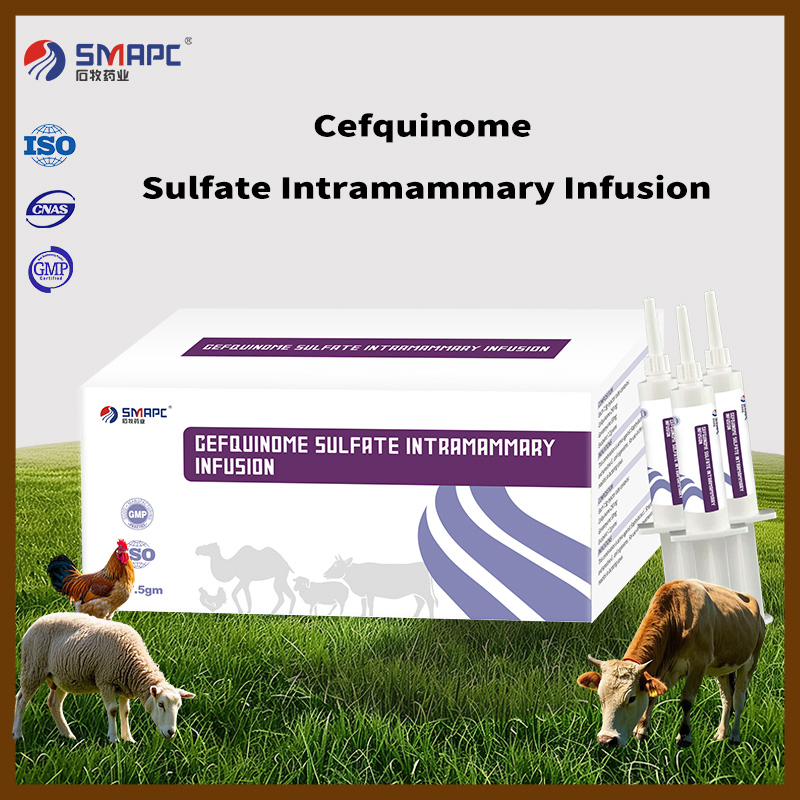
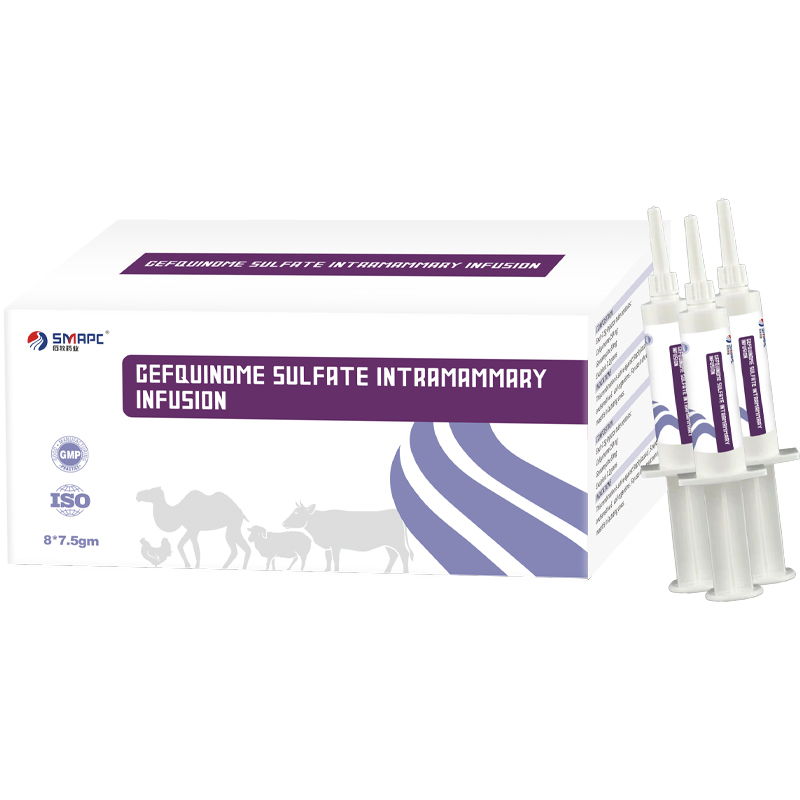
| Model NO. | Cefquinome Sulfate Intramammary Infusion |
| Type | The First Class |
| Pharmacodynamic Factors | Animal Species |
| Storage Method | Light Proof |
| Product Type | Injection / Infusion |
| Efficacy | Antibacterial |
| Packing | 100ml/Bottle |
| GMP Manufacture | Yes |
| ISO Certified | Yes |
| HS Code | 3004909099 |
| Production Capacity | 50000 Pieces/Day |
| Package Size | 45.50cm * 36.00cm * 37.50cm |
| Gross Weight | 25.000kg |
| Transport Package | Cartons |
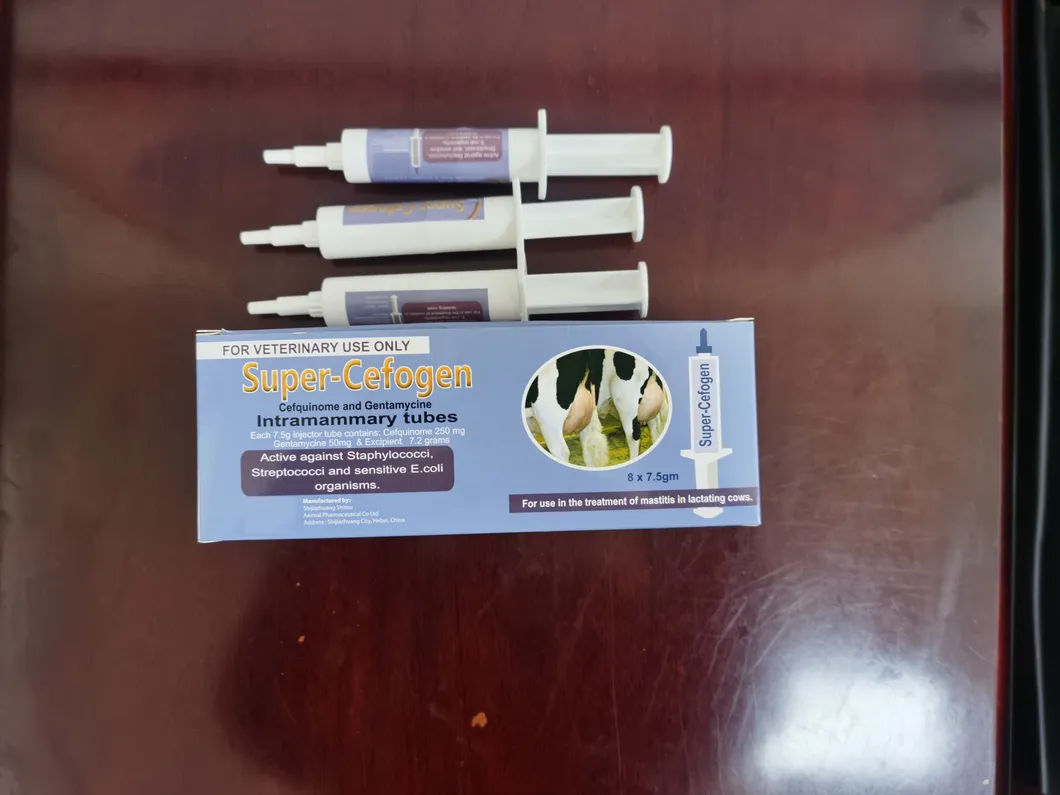

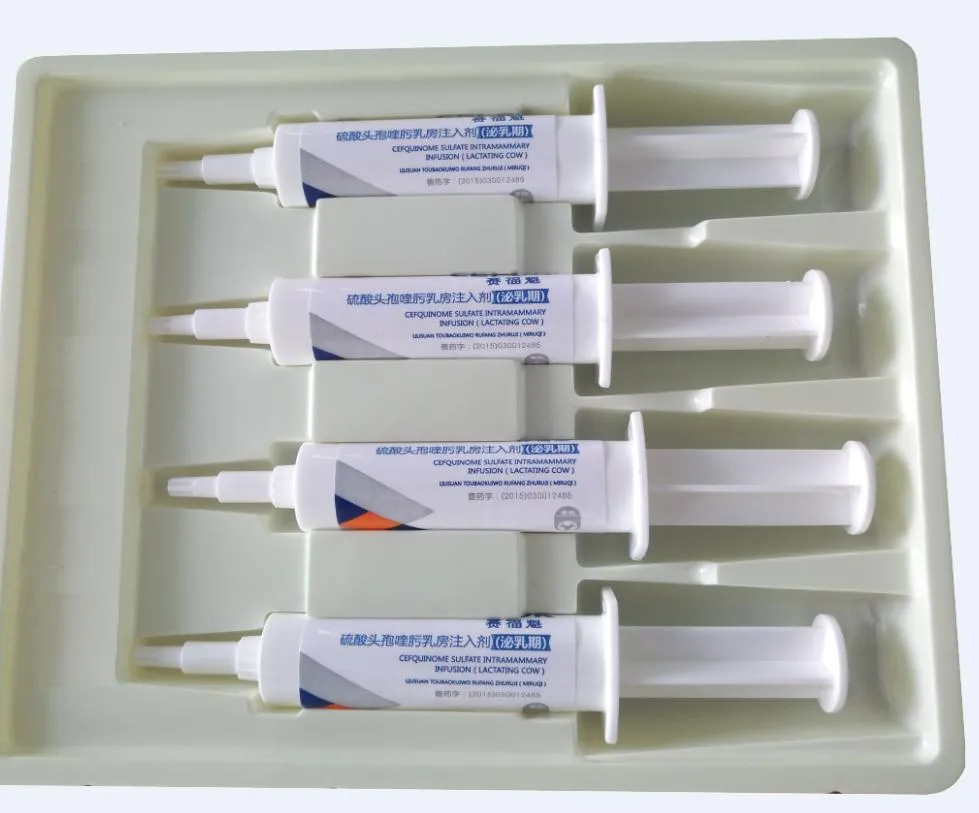
| Composition | Each syringe contains Cefquinome sulfate 75mg. |
| Pharmacological action | β-lactam antibiotics. Cefquinome is a fourth-generation cephalosporin specially for animals, showing strong antibacterial activity against common Gram-negative and Gram-positive bacteria. |
| Indication | Treating mastitis caused by sensitive bacteria (Streptococcus, Staphylococcus aureus, Escherichia coli, etc.). |
| Dosage | Intraductal injection: Lactating cow, 1 tube in each infected dairy room after milking, every 12 hours for 3 consecutive times. |
| Withdrawal Period | Milk: 96 hours. |
| Storage | Protect from light, Seal and store below 25ºC. Keep out of reach of children. |








