
1 / 5
 Vetsys Pharmaceutical
Vetsys Pharmaceutical










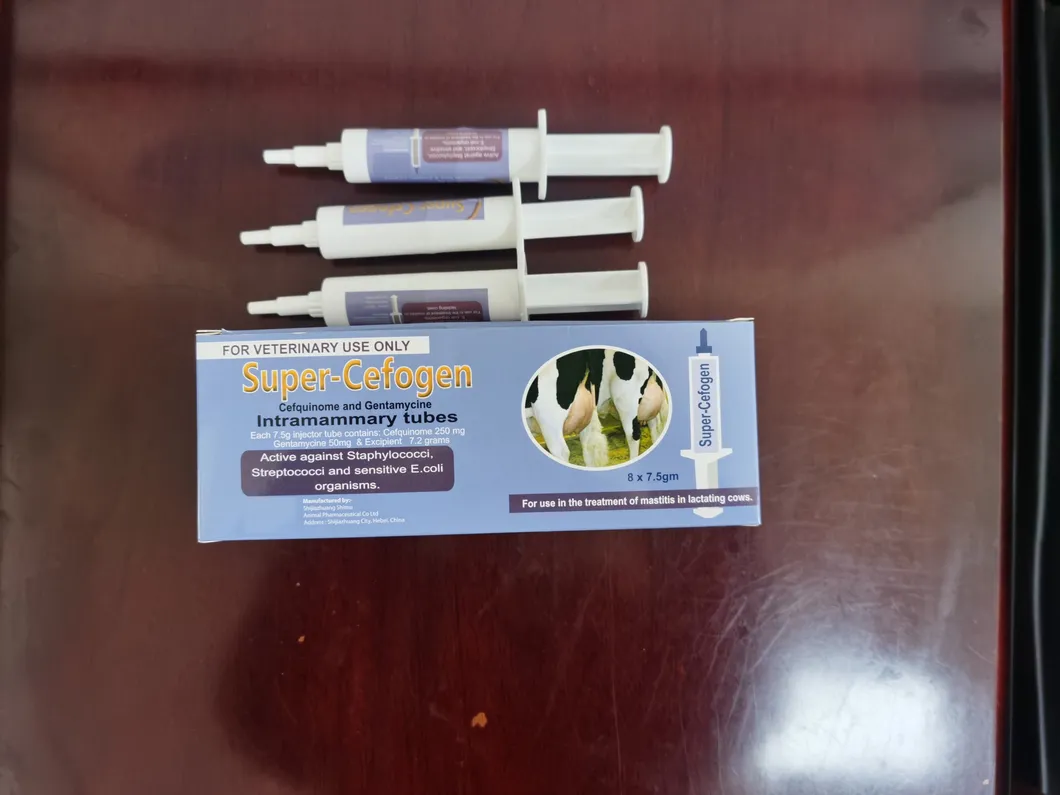

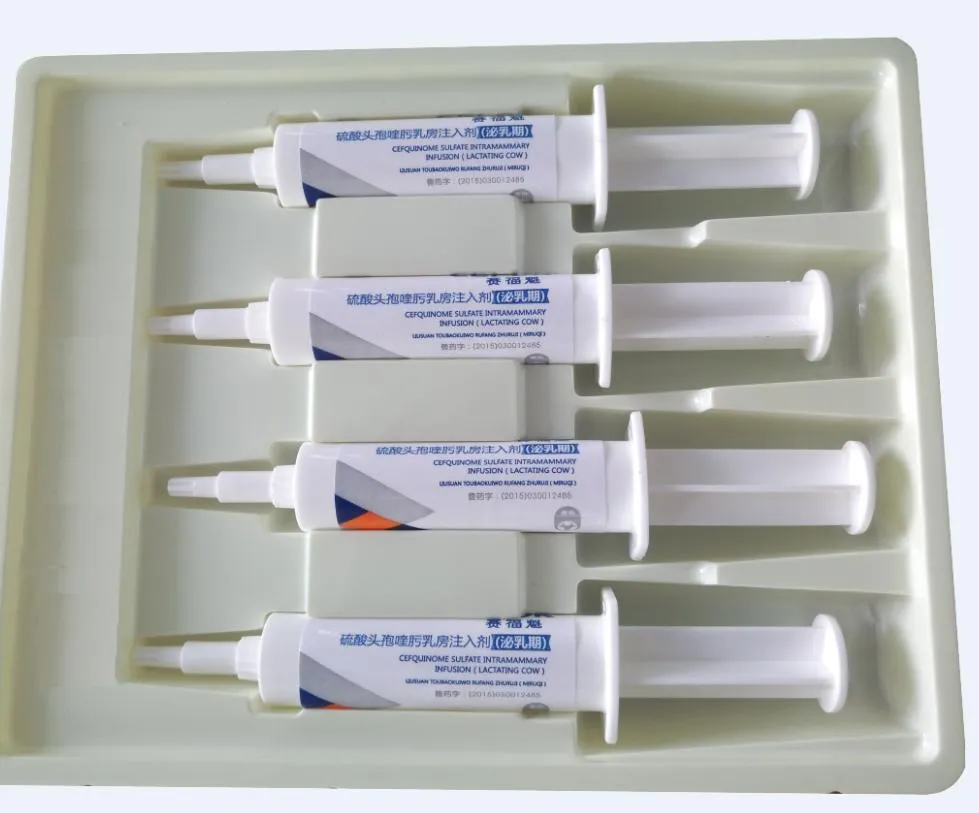
| Product Name | Cefquinome sulfate intramammary infusion (Lactating Cow) |
| Composition | Each syringe contains Cefquinome sulfate 75mg. |
| Pharmacological Action | β-lactam antibiotics. Cefquinome is the fourth generation cephalosporin antibiotic specially used for animals, which has strong antibacterial activity against common Gram-negative and Gram-positive bacteria. |
| Indication | Mainly used to treat mastitis caused by sensitive bacteria (Streptococcus, Staphylococcus aureus and Escherichia coli, etc.). |
| Dosage | Intraductal injection: Lactating cow, 1 tube in each infected dairy room after milking, inject once every 12 hours for 3 consecutive times. |
| Withdrawal Period | Milk: 96 hours. |
| Storage | Protect from light, Seal and store below 25ºC. Keep out of reach of children. |








