iBasic Information
| Model NO. |
Cefquinome Gentamycin Sulfate Intramammary Infusion |
| Type |
The First Class |
| Dosage Type |
Intramammary Infusion |
| GMP / ISO |
Yes / Yes |
| Specification |
5g/vial; 5vials/box |
| Production Capacity |
5,000,000 / Day |
| Origin |
China |
📦Packaging & Delivery
| Package Size |
45.50cm * 36.00cm * 37.50cm |
| Package Gross Weight |
25.000kg |
| Transport Package |
5g/Vial, 5vials/Box |
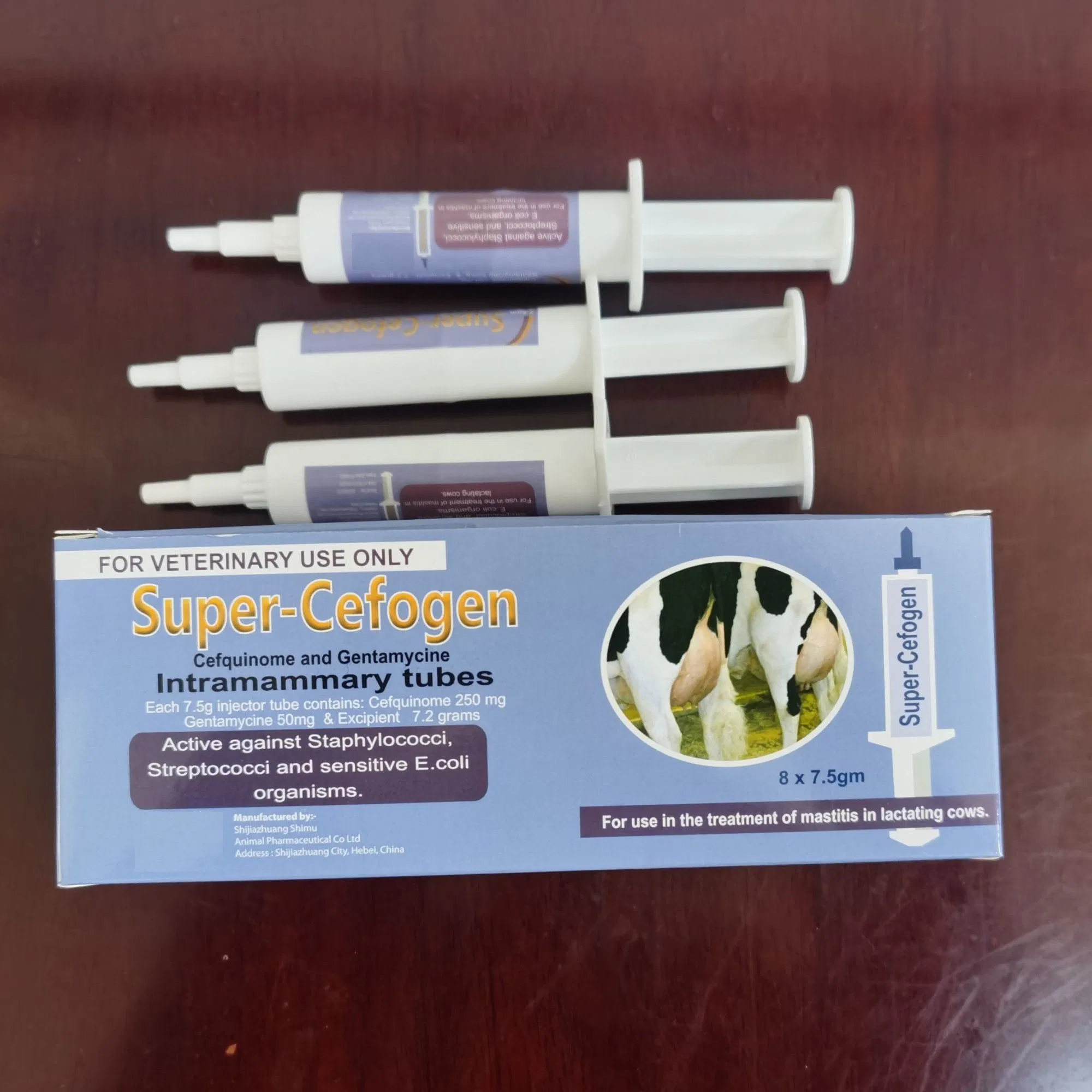
★Product Description
Composition:
Each 7.5g injector tube contains:
- Cefquinome: 250 mg
- Gentamycine: 50 mg
Indications:
Active against Staphylococci, Streptococci, and sensitive E.coli organisms. For use in the treatment of mastitis in lactating cows.
📜Certifications & Quality
- Veterinary Medicine GMP Certificate & Production License
- ISO9001 Quality Management System
- ISO14001 Environmental Management System
- ISO45001 Occupational Health and Safety Management System
- Top Tier Veterinary Medicine Enterprise
?Frequently Asked Questions
Do you have GMP certification?
Yes, the first GMP certification was successfully obtained in 2005, and the latest GMP standard was updated in 2022.
How does your factory control product quality?
Our company prioritizes quality through a dedicated Quality Management Department consisting of QA and QC units. The QC central laboratory is equipped with advanced inspection instruments to ensure no unqualified materials or intermediate products enter the next process.
What management certificates do you hold?
We hold a Manufacturing License and GMP. Additionally, we have passed ISO9001, ISO 14001, and ISO45001 certifications.
Can you provide documentation for product registration?
Yes, we have a professional registration team. We can provide documents including FSC, COPP, COA, Analysis Methods, SMF, Stability Reports, and Instructions.
Is it possible to visit your manufacturing facility?
Every year, many international customers visit our facilities. You are welcome to visit our veterinary medicine factory to see our production processes firsthand.
What is your production capacity?
We have a robust manufacturing capability with a production capacity of up to 5,000,000 units per day.




 Vetsys Pharmaceutical
Vetsys Pharmaceutical
















