


1 / 5
 Vetsys Pharmaceutical
Vetsys Pharmaceutical










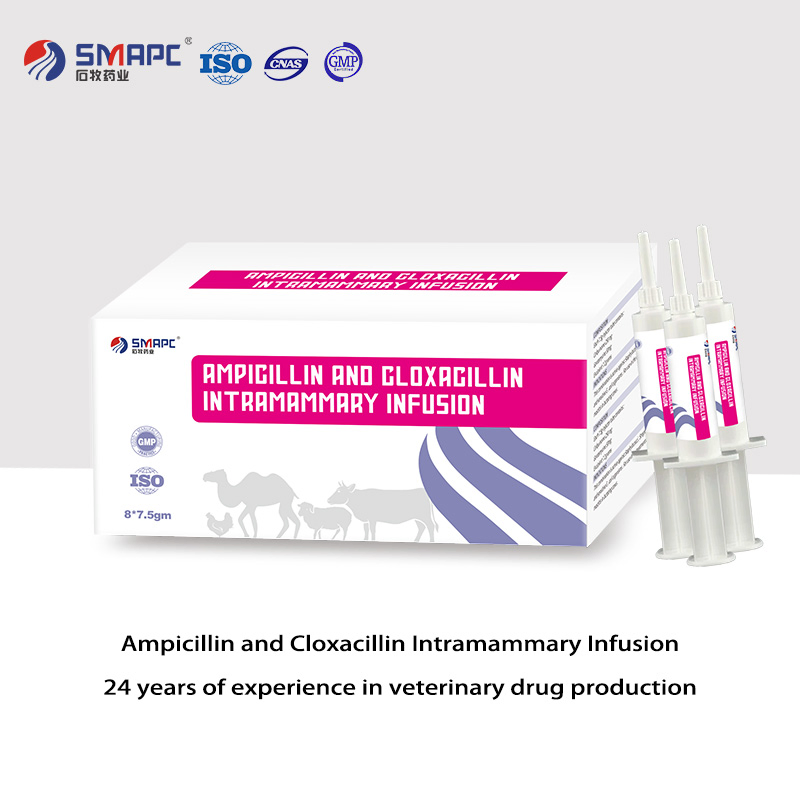
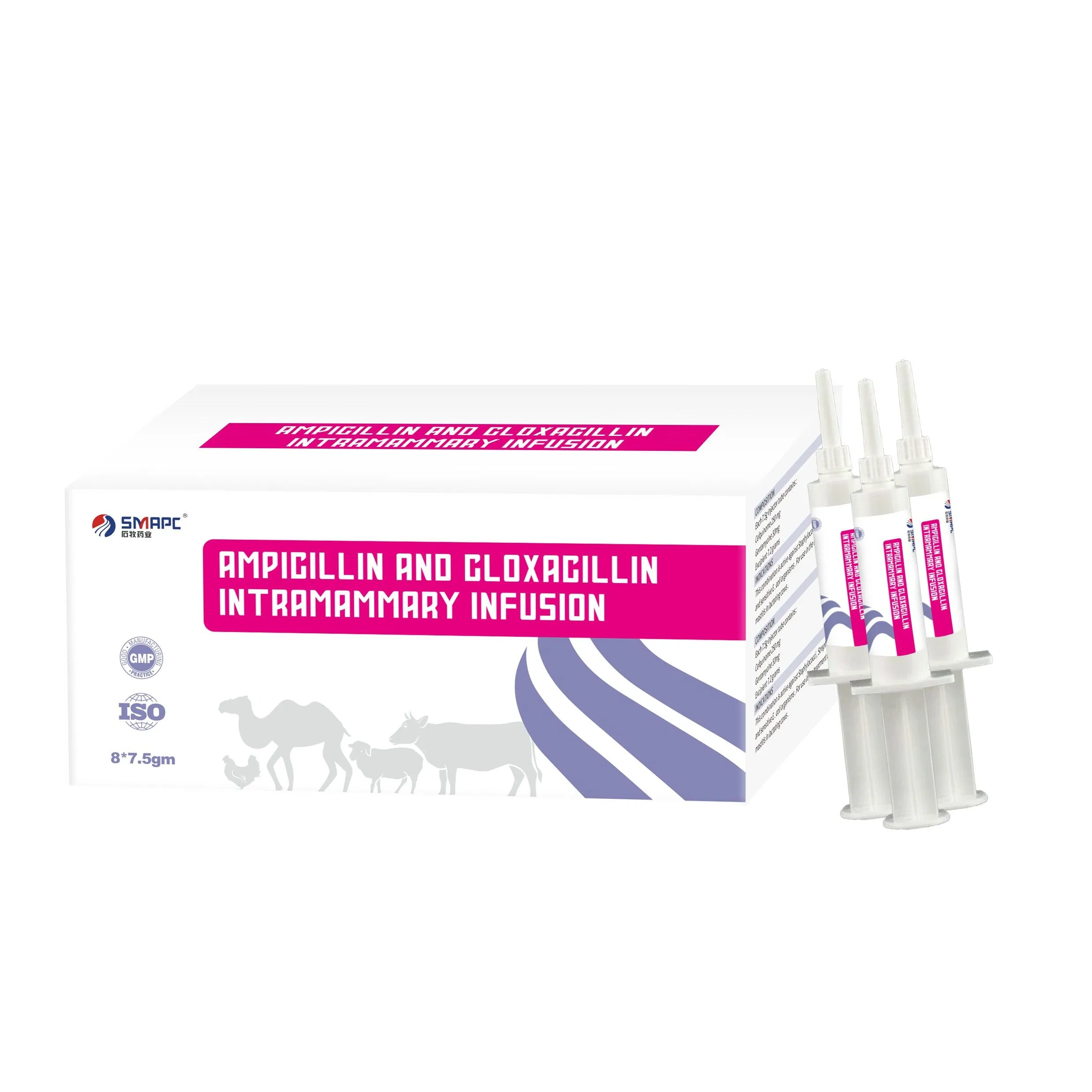
| Model NO. | Ampicillin and Cloxacillin Intramammary Infusion |
|---|---|
| Type | The First Class |
| Product Type | Intramammary Infusion |
| Efficacy | Antibacterial |
| GMP Manufacture | Yes |
| ISO Certified | Yes |
| Specification | 100ml/bottle |
| Origin | China |
| Production Capacity | 50000 Pieces/Day |
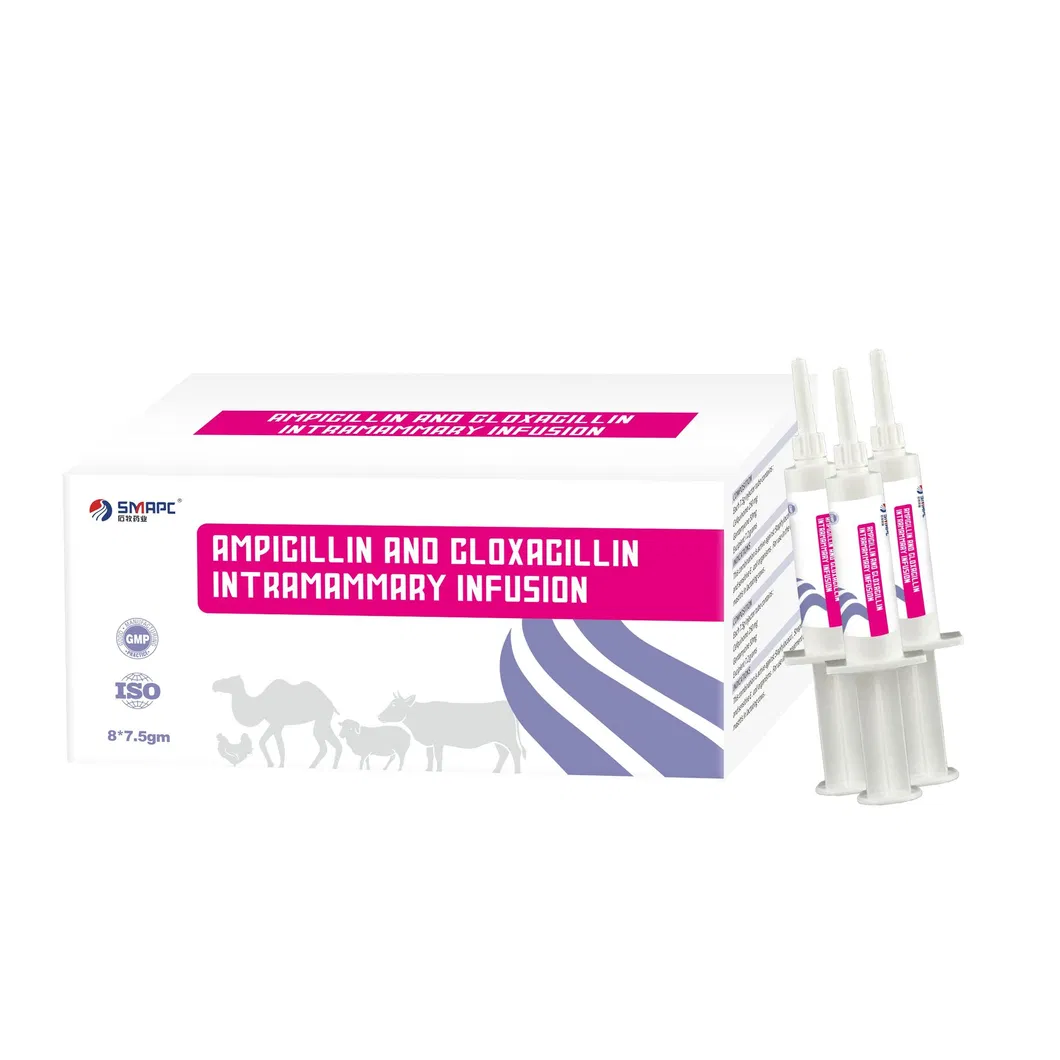

| Composition | Per syringe: Ampicillin 75mg, Cloxacillin 200mg |
|---|---|
| Indications | Mainly used to treat breast infections caused by Gram-positive and Gram-negative bacteria. |
| Dosage (Lactating Cow) | After-milking, 5.0g injected into each milk chamber every 12 hours based on condition. |
| Withdrawal Period | Cow: 28 days; Milk: 60 hours (48 hours of abandoned milk). |
| Storage | Seal and store in a cool, dark place below 30ºC. |
| Shelf Life | 2 Years |
| Packaging | 5g/syringe, 10 syringes/box |






Albendazole Tablet: Broad-spectrum anthelmintic for tapeworms, roundworms, and hookworms.
Multivitamin Tablet: Comprehensive nutritional support for growth and immune function.
Tylosin Tartrate Injection: Effective for respiratory infections, mastitis, and pneumonia.
Amoxicillin Soluble Powder: Used for urinary tract and soft tissue infections in all species.
Povidone Iodine 10% Solution: High-efficiency disinfectant for bacteria, viruses, and fungi.