



 Vetsys Pharmaceutical
Vetsys Pharmaceutical










| Model NO. | Premix |
|---|---|
| Storage Method | Prevent Throwing Expired Veterinary Drugs |
| Sample | Yes |
| Factory | Yes |
| GMP | Yes |
| Purity | No Less Than 96% |
| Transport Package | 25kg/Drum |
| Specification | 25kg/bag |
| Origin | China |
| HS Code | 2932209090 |
| Production Capacity | 250ton/Year |

🌟 Good Water Solubility & Absorption: The advanced water-soluble design is more conducive to the intestinal absorption of animals. It can be completely dissolved in water within 5-10 minutes.
🛡️ No Drug Resistance: This product has been used globally for over 50 years without significant resistance. No similarity to other antibiotics means no cross-resistance issues.
🎯 Professional Coating: Adopting the latest international coating technology, particles are even and mix easily in feed, ensuring consistent concentration and excellent palatability.
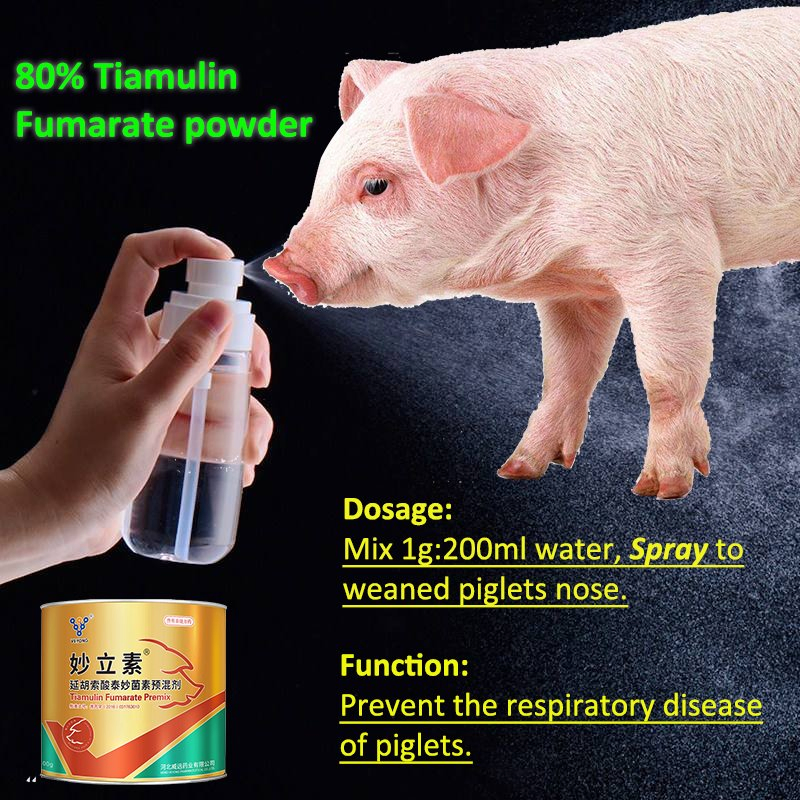
🔄 Flexible Administration: Can be used via mixing, drinking water, spraying, nose drops, or injection for versatile prevention and treatment.
| Mixing Category | Usage and Administration | Main Function |
|---|---|---|
| Boar | Mix 150g with 1000kg feed for 7 days. | Reduce respiratory pathogens; prevent spread to piglets. |
| Piglet | Mix 150g with 1000kg feed for 7 days. | Reduce weaning stress and respiratory disease incidence. |
| Fattening Pig | Mix 150g with 1000kg feed for 7 days. | Prevent respiratory diseases and swine ileitis. |
💧 Drinking Water: 50 grams per 500 kilograms of water for respiratory diseases.
🔬 Ileitis Control: 150g per ton of feed or 50g per 500kg water for two weeks.
⚠️ Precautions: Do not combine with polyether antibiotics (monensin, salinomycin, etc.) to avoid poisoning. If poisoning occurs, stop immediately and treat with 10% glucose solution.
| Specification | EP; USP | Usage | Antiparasite |
|---|---|---|---|
| Raw Material | Tiamulin hydrogen fumarate | Species | Cattle, sheep, swine, camels |
| Dosage | API | Sample | Yes |
| Packing | 25KG/drum | Delivery | 7-10 days |
| Payment | T/T, D/P, D/A, L/C | ||




Established in 2002, the enterprise is a large GMP-certified veterinary drug manufacturer specializing in R&D, production, and sales of veterinary APIs, preparations, premixed feeds, and feed additives. With a dedicated team of 65 technical professionals, the company maintains an innovative R&D system for new veterinary drugs.
The production bases cover extensive areas, featuring 13 API products including Ivermectin, Eprinomectin, and Tiamulin Fumarate, alongside 12 preparation production lines. The facility operates under strict EHS (Environment, Health & Safety) management systems, holding ISO14001 and OHSAS18001 certificates. The quality management system is backed by ISO9001, China GMP, Australia APVMA GMP, and US FDA inspections.
With products exported to Europe, South America, the Middle East, Africa, and Asia (over 60 countries), the company has built long-term cooperation with internationally known pharmaceutical enterprises through excellent product quality and scientific management.



