

 Vetsys Pharmaceutical
Vetsys Pharmaceutical






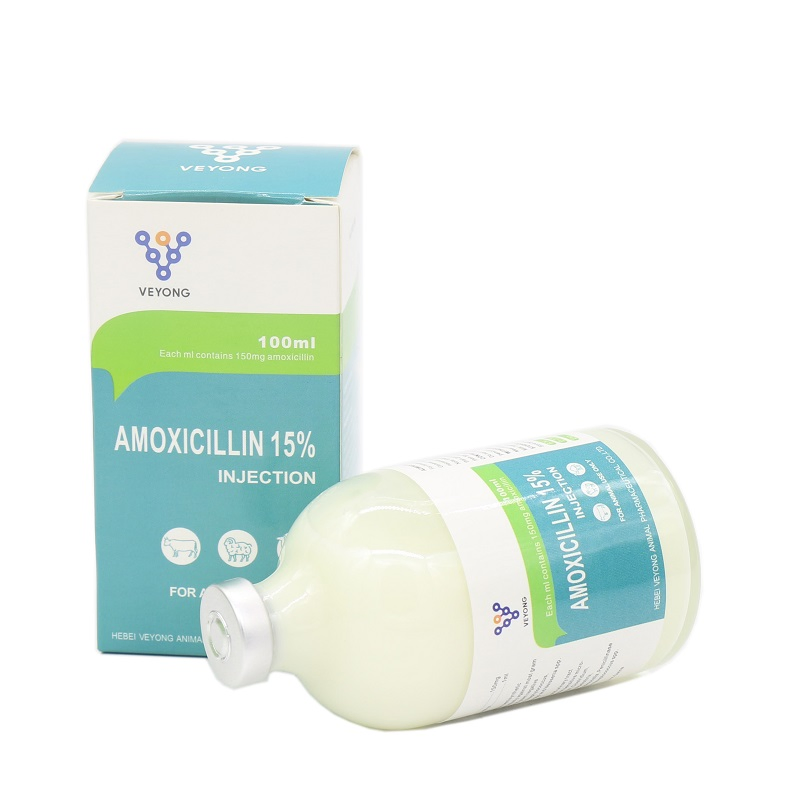
| Specification | 15% | Usage | Antibiotics |
| Raw Material | Amoxicillin | Species | Cattle, sheep, camel, chicken |
| Dosage Form | Injection | Sample | Yes |
| Packing | 50ml/vial/box | Delivery Time | 15-20 days |
| MOQ | 10000 bottles | Payment Term | T/T, D/P, D/A, L/C |
| GMP / ISO9001 | Yes / Yes | FDA / COS | Yes / Yes |
Indications:
Contains 150mg amoxicillin base per ML. Amoxicillin is primarily used for treating infections caused by gram-positive and negative bacteria susceptible to penicillin. It is suitable for diseases in poultry and livestock including fevers, loss of appetite, constipation, shortness of breath, and abdominal breath. Effective against flu, nameless fever, bacillary dysentery, acute gastroenteritis, pig's erysipelas, pneumonic plague, piglet's diarrhea, paratyphoid, E. coli infections, brucella, mycoplasma, leptospirosis, poultry cholera, chicken's dysentery, salpingitis, and mastitis.
Pharmacological Action:
Acts against gram-positive bacteria (Streptococcus, Staphylococcus, Clostridium, etc.) and gram-negative bacteria (Brucella, Salmonella, E. coli, etc.). It suppresses cell wall synthesis, causing bacteria to burst and dissolve rapidly. The bactericidal action is stronger and faster compared to ampicillin.

Usage and Administration: Intramuscularly or subcutaneously injection. For livestock: 5 - 10mg amoxicillin per 1kg body weight, once daily; or 10 - 20mg per 1kg body weight, once every two days.
Side Effects: Individual livestock may show rare allergic reactions like skin rashes or oedema.
Withdrawal Time: Slaughter: 28 days; Milk: 7 days; Egg: 7 days.




Established in 2002, this large GMP-certified veterinary drug enterprise integrates R&D, production, and sales of veterinary APIs, preparations, and feed additives. As a Provincial Technical Center, it maintains an innovative R&D system with 65 technical professionals. The production base covers 78,706 m², featuring 13 API products including Ivermectin and Oxytetracycline, alongside 12 preparation production lines.
The enterprise adheres to EHS (Environment, Health & Safety) systems, holding ISO14001 and OHSAS18001 certifications. With a complete quality management system, it has passed inspections by the US FDA and holds certificates from Australia (APVMA) and Ethiopia. Products are exported to more than 60 countries and regions across Europe, South America, the Middle East, Africa, and Asia.






